A New Era in Diabetes Care: Once-Weekly Insulin Icodec
Imagine telling a person with type 2 on daily injections:
“Instead of taking your basal insulin every day, you only need it once a week.”
That future is no longer theoretical.
With the development and FDA approvals of insulin icodec for people with type 2 diabetes, we are entering a new era of diabetes care—one that prioritizes simplicity, adherence, and person-centered innovation.
What Is Insulin Icodec?
Insulin icodec (brand name Awiqli®) is the first once-weekly basal insulin analog designed to provide steady glucose control over 7 days with a single injection.
Unlike traditional basal insulins, icodec is engineered to:
- Bind to albumin in the bloodstream, creating a circulating depot
- Release insulin slowly over the course of a week
- Maintain stable basal insulin levels with fewer peaks and troughs
This innovation reduces injections from 365 per year to just 52, a meaningful shift for many people living with diabetes.
Pharmacology Spotlight: Why Insulin Icodec Is Unique
Insulin icodec represents a significant pharmacologic advancement as a once-weekly, ultra–long-acting basal insulin formulated at U-700 concentration (700 units/mL).
This high-concentration formulation allows delivery of a full week’s basal insulin in a single injection while maintaining a manageable injection volume.
Structurally, icodec is designed with strong, reversible albumin binding, creating a circulating depot that slowly releases insulin over approximately 7 days. It has a half-life of about one week, reaching steady state after several weekly doses, which supports consistent basal coverage with minimal peak-to-trough variability.
Because of this prolonged action, dose adjustments must be made thoughtfully, as changes take longer to fully manifest. Understanding this pharmacokinetic profile is essential to safely initiating, titrating, and educating patients about this novel therapy.
Clinical Efficacy: What Does the Evidence Show?
The ONWARDS Phase 3 clinical program (≈4,000 participants with type 2 diabetes) demonstrated:
- A1C reduction comparable to once-daily basal insulin
- Non-inferiority to standard basal insulins like degludec or glargine
- Similar risk of hypoglycemia
- Slightly increased time in range
For clinicians, this means:
👉 Similar outcomes
👉 With fewer injections
👉 And potentially happier patients
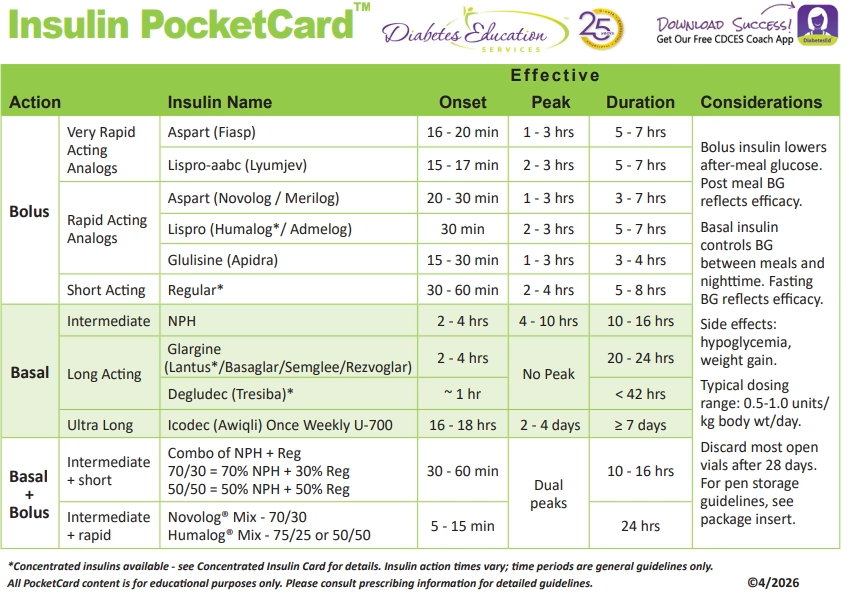
Download FREE Insulin PocketCard
Our Insulin PocketCard Now includes icodec insulin.
Download your own ePocketCard Today here! or
Download our CDCES Coach App which has the latest ePocketCard version!
Safety Considerations: What Should We Watch For?
1. Hypoglycemia Risk and Considerations
- The effect of insulin icodec is highest at Days 2-4 of the weekly dosing cycle, increasing the risk of hypoglycemia on these days. See Awiqli Monograph
- There are still concerns about increased hypoglycemia risk for people with type 1 diabetes. For that reason, the FDA has not approved insulin icodec for use in people with type 1 diabetes at this time.
- For those with type 2 diabetes, the risk of hypoglycemia is comparable to those on daily bolus injections.
- Diabetes healthcare professionals are encouraged to review the signs of hypoglycemia and importance of quickly consuming carbs to raise glucose levels.
- Making sure individuals on icodec have a meter or CGM to monitor glucose response is imperative for safety, especially when making dose adjustments.
- If someone becomes acutely ill and is not eating or needs to fast for a procedure, there is less flexibility to immediately adjust the dose because of the ultra long insulin half-life.
- Health care teams will need to have protocols for how to treat patients admitted to hospital on weekly insulin to ensure they are not giving additional basal insulin when the weekly insulin is still in their system.
2. Dosing & Titration Complexity – See Package Insert for Dosing Info
- When initiating insulin icodec, dosing is typically based on the person’s current total daily basal insulin dose, with the goal of maintaining equivalent weekly insulin exposure.
- A practical approach is to multiply the existing daily basal dose by 7 to estimate the initial once-weekly dose, followed by careful titration based on fasting glucose trends.
See Package Insert for Dosing Info
- For individuals who are insulin-naïve, a standard starting dose (e.g., 70 units once weekly in clinical trials) may be used, with adjustments made weekly or every few weeks depending on glycemic response.
- Because of icodec’s long half-life and delayed steady state, dose changes take longer to fully manifest, so clinicians should avoid making rapid or frequent adjustments.
- As always, individualization is key—consider factors such as hypoglycemia risk, renal function, and patient ability to monitor glucose when determining and adjusting the dose.
3. Dose Adjustment in Altered Kidney Function – See Package Insert for Dosing Info
The following recommendations based on kidney function are suggested.
- eGFR of 50 or greater – no dosage adjustment needed.
- eGFR of 10-50 – administer 75% of the usual starting dose.
- eGFR less than 10 – administer 50% of usual dose.
4. Missed Doses – See Package Insert for Dosing Info
The following recommendations for missed doses are suggested:
- If 3 or fewer days until next regularly scheduled dose: Administer missed dose as soon as possible. Reset weekly dosing schedule to start one week later.
- If 4 or more days until next regularly scheduled dose: Administer missed dose as soon as possible. Continue current weekly dosing schedule.
5. Insulin Storage and Dispensing Information - See Cheat Sheet for more Info
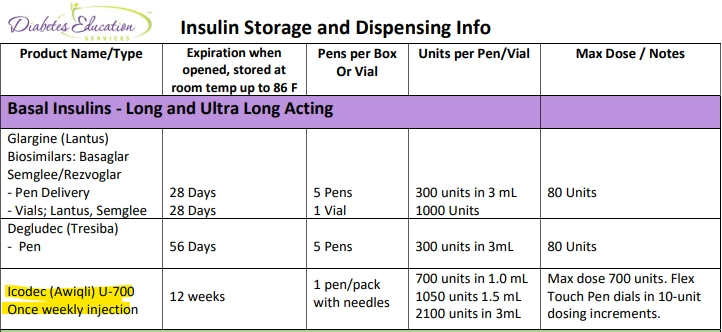
Icodec insulin is available in 3 different pen sizes and has a 12 week shelf life at room temperature. Pens can be dialed in 10 unit increments with a maximum dose of 700 units.
Download our FREE Insulin Storage and Dispensing Info Cheat Sheet below as a helpful reference.
Who Might Benefit Most from Insulin Icodec?
Insulin icodec may be especially valuable for:
- People with type 2 diabetes who struggle with taking daily injections.
- Individuals experiencing treatment fatigue
- Those who would benefit from simplified regimens
- Re-engaging individuals hesitant to start insulin
This is where your role as a healthcare professional becomes even more powerful—matching the right therapy to the right person.
Final Thoughts
Insulin icodec represents a transformational step forward in diabetes management. While ongoing evaluation is needed—especially regarding safety and real-world use—this therapy has the potential to reshape how we approach basal insulin. Novo Nordisk plans to launch and release the prefilled icodec (Awiqli) FlexTouch® pens in the US in the second half of 2026.
As healthcare professionals, staying current with these advancements allows us to:
- Provide evidence-based recommendations
- Support informed decision-making
- And most importantly, meet people where they are
Want to Stay Current on Innovations Like This?
Join us as we break down the latest in diabetes care, guidelines, and emerging therapies— you can bring confidence, clarity, and compassion to your clinical practice by joining our
Virtual DiabetesEd Training Conference.
Virtual DiabetesEd Training Conference

Why Should I Attend?
Join national experts including Dr. Diana Isaacs (Cleveland Clinic), Beverly Thomassian (30+ years of experience), and Christine Craig for high-impact, virtual learning—no travel required.
✔ Learn from National Experts — Anywhere
Get the same expert-level instruction you’d receive in person, delivered live to your home or office.
✔ Interactive & Flexible
- Live Q&A and real-time case discussions
- Direct engagement with faculty
- Full session recordings with 1-year access if you can’t attend live
What's Included?
- 19.5 CEs from the 3-day interactive conference + 17+ bonus CEs from 11 on-demand courses. (30+ total CEs are AMA PRA Category 1™, ACPE, ANCC, CDR accredited)
- Less than $15 per credit!
- Expert-led sessions: ADA Standards, medications, insulin therapy, behavior change, CGM, medical nutrition therapy
- Interactive Q&A with expert faculty during live sessions
- 1-year access to all recordings that you can watch on your schedule
- Study material for CDCES and BC-ADM exam prep
Set Yourself Up With Certification Success
Get exam-ready with confidence.
Course credits through AMA PRA Category 1 Credits™, ACPE, ANCC, and CDR!
Full accreditation details are available on the registration page

Our CDCES Boot Camp Online Prep Bundle is a comprehensive, high-impact program built specifically for healthcare professionals preparing for the Certified Diabetes Care and Education Specialist (CDCES) exam who want to level up their clinical knowledge and skills.

This evidence-based study bundle is a comprehensive BC-ADM Boot Camp designed for advanced-level healthcare professionals preparing for the Board Certified in Advanced Diabetes Management (BC-ADM) exam and will also provide you with state-of-the-art information to level up your clinical practice.
Hear What Our Community Has to Say