
For last week’s practice question, we quizzed test takers on Mounjaro, a new medication. 82% of respondents chose the best answer. We want to share this important information, so you can pass it on to people living with diabetes and your colleagues, plus prepare for exam success!
Before we start though, if you don’t want any spoilers and haven’t tried the question yet, you can answer it below: Answer Question
Question: The FDA recently approved the new ‘twincretin” dual incretin hormone therapy called Tirzepatide (Mounjaro). The provider wants to start a person with type 2 diabetes on this new medication and asks for your consultation. The person’s current medications for type 2 diabetes include metformin, empagliflozin (Jardiance), semaglutide (Ozempic), and Pravachol. What is the best response to the provider?
Answer Choices:
- Inform provider that Tirzepatide (Mounjaro) is contraindicated in people taking SGLT-2 Inhibitors.
- Recommend stopping the semaglutide (Ozempic) before starting the Tirzepatide (Mounjaro).
- Reinforce the importance of prescribing a CGM before starting Tirzepatide (Mounjaro).
- Request provider also prescribes Glucagon rescue med in case of severe hypoglycemia.
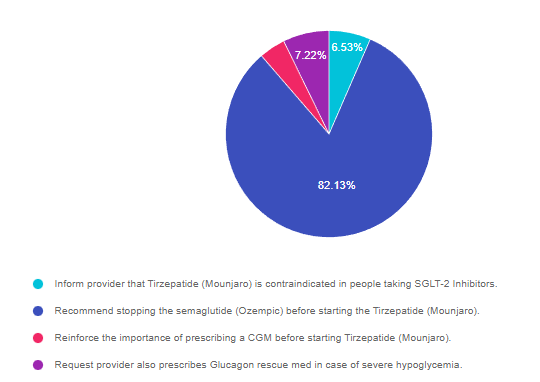
As shown above, the most common choice was option 2, the second most common answer was option 4, then option 1, and finally 3.
Getting to the Best Answer
Answer 1 is incorrect. 6.53% chose this answer, “Inform provider that Tirzepatide (Mounjaro) is contraindicated in people taking SGLT-2 Inhibitors.” According to the Package Insert, Tirzepatide can be used in combination with SGLT-2 Inhibitors and other approved diabetes medications. However, since Tirzepatide is a combination of GLP-1 and GIP Incretins, it is not recommended to use in combination with a GLP-1 RA or the DPP-IV class of medications. Read more on our blog here.
Answer 2 is correct. 82.13% of you chose this answer, “Recommend stopping the semaglutide (Ozempic) before starting the Tirzepatide (Mounjaro).” YES, GREAT JOB! Since Tirzepatide is a combination of GLP-1 and GIP Incretins, it is not recommended to use in combination with a GLP-1 RA or the DPP-IV class of medications.
Answer 3 is incorrect. 4.12% of respondents chose this answer, “Reinforce the importance of prescribing a CGM before starting Tirzepatide (Mounjaro).” Since this person is not on insulin or any medications that would cause low blood sugar, a CGM is not required to monitor blood sugars. Blood glucose monitoring combined with regular A1C testing will be important to determine the efficacy of this new “twincretin”. Read more on our blog here.
Finally, Answer 4 is incorrect. 7.22% chose this answer, “Request provider also prescribes Glucagon rescue med in case of severe hypoglycemia.” Since this person is not on insulin or any medications that would cause low blood sugar, a glucagon rescue med is not required to treat severe hypoglycemia. Blood glucose monitoring combined with regular A1C testing will be important to determine the efficacy of this new “twincretin”. Read more on our blog here.
Want to learn more about this question?
New Dual Incretin Injectable – “TwinCretin”
The FDA just approved a novel, dual incretin hormone therapy called Tirzepatide (Mounjaro).
This new twin incretin injectable includes not only a GLP-1 Receptor Agonist, but also a Glucose-dependent insulinotropic polypeptide (GIP), which magnifies the therapeutic effectiveness. The SURPASS studies indicate that study participants experienced an A1C drop of up to 2.5% and weight loss of up to 10kg or more.
Want to learn more about Diabetes Care? View our
Virtual DiabetesEd Specialist Conference
30+ CEs
Airs October 12-14th, 2022

Whether you are new to diabetes or a seasoned expert, you’ll benefit from this virtual conference with the latest research plus critical content that you can immediately apply to your clinical practice.
Download Course Flyer | Download Schedule
If you are seeking a state-of-the-art review of current diabetes care, this course is for you. Our team has been fine-tuning this course for over fifteen years, and we know what you need. This program can also be a great addition to your CDCES or BC-ADM exam study plan.
Join us LIVE for this Virtual Course and enjoy a sense of community!
Team of expert faculty includes:
- Diana Isaacs, PharmD, BCPS, BC-ADM, BCACP, CDCES – Educator of the Year, 2020
- Coach Beverly Thomassian, RN, MPH, CDCES, BC-ADM
- Ashley LaBrier, MS, RD, CDCES, Diabetes Program Coordinator
Two Registration Options
Virtual DiabetesEd Specialist Conference Deluxe | 30+ CEs
Deluxe Option for $499: Virtual Program includes:
- Q & A Session with the instructor after each webinar.
- LIVE Presentations by our team of experts.
- State-of-the-art review of current diabetes care and technology.
- Resources for each session.
- Access to free podcasts and video recordings within a week of each live session for one year.
Deluxe Version includes Syllabus, Standards and Swag*:
- Diabetes Educator Course 2022 Syllabus Hard Copy – over 100 pages -This spiral-bound workbook contains the printed version of all of the instructor’s slides.
- ADA 2022 Standards of Care Book -The ADA Standards of Medical Care in Diabetes is a key resource for healthcare professionals involved in diabetes care, education, and support.
- DiabetesEd Services highlighters, Medication PocketCard, Tote Bag and Pen
Virtual DiabetesEd Specialist Conference Basic | 30+ CEs
Basic Option for $399: Virtual Program includes:
- Q & A Session with the instructor after each webinar.
- LIVE Presentations by our team of experts.
- State-of-the-art review of current diabetes care and technology.
- Resources for each session.
- Access to free podcasts and video recordings within a week of each live session for one year.
Don’t worry if you can’t make it live. Your registration guarantees access to the recorded version in the Online University.
All hours earned count toward your CDCES Accreditation Information
Sign up for Diabetes Blog Bytes – we post one daily Blog Byte from Monday to Friday. And of course, Tuesday is our Question of the Week. It’s Informative and FREE! Sign up below!
The use of DES products does not guarantee the successful passage of the CDCES exam. CBDCE does not endorse any preparatory or review materials for the CDCES exam, except for those published by CBDCE.




