 Once weekly basal insulin, icodec, failed to be approved for use by people with type 1 diabetes by the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee by a vote of 7-4. The significantly increased risk for hypoglycemia on days two through four after administration outweighed its potential benefits. The committee also noted that icodec’s risk of hypoglycemia is higher than the basal insulin degludec, which is commonly used and has a better safety profile. Other committee members expressed concerns that approving icodec based on inadequate data could discourage further trials that are needed to ensure its safe use.
Once weekly basal insulin, icodec, failed to be approved for use by people with type 1 diabetes by the FDA’s Endocrinologic and Metabolic Drugs Advisory Committee by a vote of 7-4. The significantly increased risk for hypoglycemia on days two through four after administration outweighed its potential benefits. The committee also noted that icodec’s risk of hypoglycemia is higher than the basal insulin degludec, which is commonly used and has a better safety profile. Other committee members expressed concerns that approving icodec based on inadequate data could discourage further trials that are needed to ensure its safe use.
In an effort to secure approval, the applicants for icodec suggested the following actions to improve the safety profile of this novel weekly insulin.
Proposed mitigating actions to reduce hypoglycemia risk by the applicant included;
- icodec only be used for people using a CGM
- individuals with hypoglycemia unawareness would not be candidates for this once weekly insulin
- labeling alternative insulin dose titration strategies to reduce the risk of hypoglycemia (e.g., reducing the bolus insulin dose by approximately 30% between days 2 to 4 after each weekly insulin icodec injection).
- Indicating insulin icodec only for patients with a low % coefficient of variation (%CV) since the subgroup of patients with low glycemic variability, as defined as percent coefficient of variation (%CV<36%), had hypoglycemia risk comparable to the entire cohort of patients on insulin degludec (i.e.,with any %CV).
- Recommending that individuals who experience recurrent hypoglycemia switch to other insulin treatment options
- Providing prescriber and patient training materials to help maximize benefit-risk.
Even though mitigating actions were suggested to decrease this hypoglycemia risk during this two day peak, the FDA panel members still deferred approval, citing safety concerns due to the significant hypoglycemia risk and need for more data..
How would people living with type 1 diabetes benefit from once weekly insulin?
Surprisingly, about one third of people living with type 1 diabetes are still managing blood sugars with multiple daily injections. This is especially true for people living in under resourced communities and people of color living with type 1 diabetes. Due to barriers and social determinants of health, in addition to struggling with multiple daily injections, they are also less likely to use continuous glucose monitors or check blood sugars on a regular basis. Although, once a week insulin seems ideal for individuals who may be experiencing a variety of barriers to injecting daily insulin, the main issue is the increased risk of hypoglycemia during days 2-4 when icodec is peaking coupled with limited access to glucose monitoring.
In addition, consistent injected insulin therapy in adults with type 1 diabetes was reported to be relatively low (52.6%, 95% confidence interval[CI]: 37.4 to 67.9%) in data from a meta-analysis of eight clinical trials. The probability of missing at least one daily basal insulin dose over any 14-day period is estimated to be 22% (95% CI: 10 to 40%).
Among individuals with type 2 diabetes, using a daily basal insulin, a once weekly basal insulin would reduce the number of insulin injections from 365 per year to 52 per year. In a recent study, 91% of people with type 2 diabetes and 89% of providers had a positive view of taking basal insulin once weekly.
Among individuals with type 1 diabetes, who rely on a basal bolus regimen, a once weekly basal insulin would reduce the number of insulin injections from approximately 28 per week to 22 per week. For those with type 1 diabetes, there is no research to date that evaluates whether a once weekly basal insulin would be preferred over other basal insulin options, or whether use would result in improved adherence and glycemic control.
ONWARDS 6 Study Results – Using icodec for people living with Type 1
In ONWARDS 6, weekly insulin icodec was noninferior (but not superior) to daily insulin degludec and was associated with 48 to 89% more level two and three hypoglycemia at Week 26, depending on the method of analysis. The highest risk period for hypoglycemia with insulin icodec coincides with its peak glucose-lowering effect which occurs on days 2 to 4 following each weekly injection. There were also more hypoglycemia-related serious adverse events reported among patients randomized to insulin icodec compared to insulin degludec.
Thus, in the only study conducted in participants with type 1 diabetes, insulin icodec was observed to have a higher risk of clinically meaningful hypoglycemia, in the absence of a lower A1C. Hypoglycemic episodes reported with insulin icodec and insulin degludec in ONWARDS 6 were of the same nature in terms of duration, management, and recovery.
What is Insulin icodec?
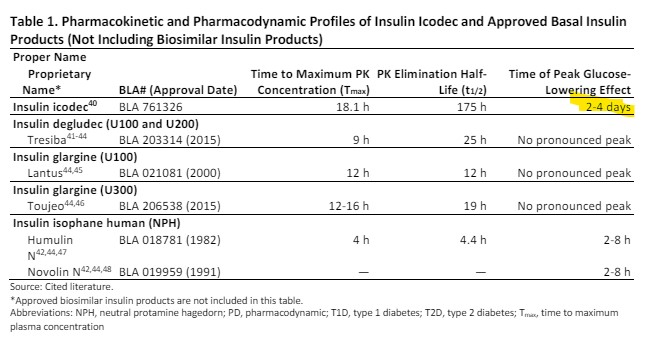
Insulin icodec is an acylated long-acting human insulin analog produced by a process that includes expression of recombinant DNA in yeast (Saccharomyces cerevisiae), followed by chemical modification. In addition to amino acid sequencing changes, a C20 fatty-acid side chain has been added to the peptide backbone via the amino group in the side chain at Lys(B29). When insulin icodec is injected, the C20 fatty acid sidechain derivative binds strongly, but reversibly, to endogenous albumin, which results in decreased renal clearance and protection from metabolic degradation, and consequently prolonged pharmacodynamic activity.
Insulin icodec is a proposed insulin analog with a prolonged duration of action intended to support once weekly (QW) subcutaneous administration. Thus, insulin icodec reduces treatment burden in type 1 diabetes, by reducing the number of basal insulin injections in comparison to daily basal insulins.
However,basal insulin icodec does not have a peakless time-action profile throughout the dosing interval (see chart below).
In conclusion, it seems certain that the manufacturers of insulin icodec will be seeking approval for this once weekly insulin for people living with type 1 and type 2 diabetes in the future. Stay tuned for more insulin updates with our monthly newsletter.
Information from this article was obtained from review of the FDA Presentation Document and Slides, May 24, 2024.
Accreditation: The Diabetes Educator Live Course is approved for 26 Contact Hours for nurses and CA Pharmacists and 21 CPE, Level III for RDs. Provider is approved by the California Board of Registered Nursing, Provider # 12640 and Commission on Dietetic Registration (CDR), Provider # DI002. Need hours for your CDCES? We have great news. This program is accredited by the CDR so all hours of instruction can be used to renew your CDCES regardless of your profession. **
The use of DES products does not guarantee the successful passage of the diabetes certification exams. CBDCE & ADCES does not endorse any preparatory or review materials for the certification exams, except for those published by CBDCE & ADCES.
**To satisfy the requirement for renewal of certification by continuing education for the Certification Board for Diabetes Care & Education (CBDCE), continuing education activities must be applicable to diabetes and approved by a provider on the CBDCE List of Recognized Providers (www.cbdce.org). CBDCE does not approve continuing education. Diabetes Education Services is accredited/approved by the Commission of Dietetic Registration which is on the list of CBDCE Recognized Providers.